Dear New World Investor:
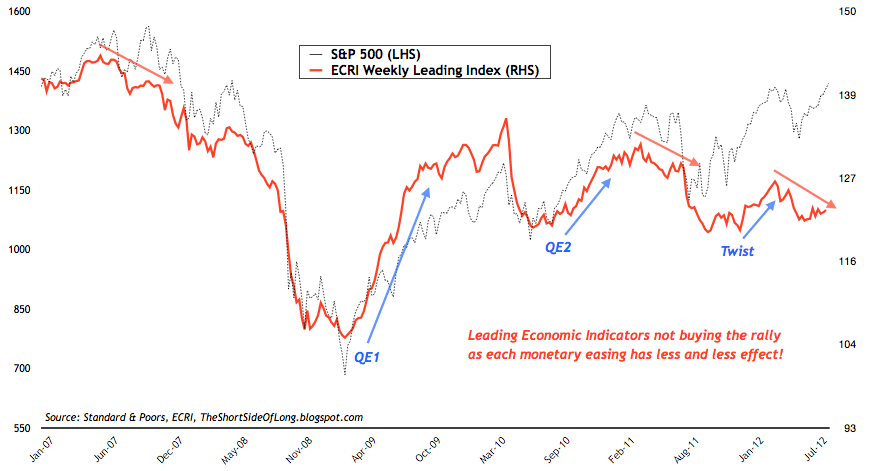
Here we are at the end of August, the time I expected to be selling some stocks to hunker down for the recession I expect next year. The seasonality pattern in Presidential years says to sell:
The Economic Cycles Research Institute Weekly Leading Indicator says to sell:
But we can’t sell. We have to wait for an academic who has never managed so much as a Burger King to speak at 10:00am ET on Friday, because we might miss a monster rally as he floods the U.S. economy with paper dollars and credit. Or not.
I expect Bernanke tomorrow to repeat that more quantitative easing should be expected if economic conditions do not show substantial improvement. He also will say that he is dissatisfied with the employment situation, and may mention a laundry list of things the Fed could do: extend Operation Twist, buy more mortgage-backed securities, buy more bonds, implement a negative interest rate policy (NIRP) tied to new lending, stop paying interest on bank reserves held at the Fed, and so on. It would quiet those parroting the “out of bullets” argument.
Then next Thursday, September 6, the European Central Bank will lower its main lending rate by 25 or 50 basis points to 0.25% or 0.50%. ECB President Mario Draghi canceled his Saturday speech at Jackson Hole, so something bigger may be brewing. At the least, I expect him to repeat that he has the authority to buy sovereign debt if not doing so would “hamper the functioning of the monetary policy transmission channel.”
As I said last week: “The next day we get the August employment report, which should be weak due to the math of seasonal adjustments. That gives the Fed cover to announce a new quantitative easing program on September 13. All of this would goose the stock market and precious metals for a while, until uncertainties about a war in Iran, the fiscal cliff, a U.S. recession, the euro crises and the election reassert themselves – and we will take protective steps then.”
New Buy Recommendation: Sequenom (SQNM)
With the market at these levels and facing a possibly deep recession next year, there are very few stocks that are positioned to go up 200% to 500% while offering relatively low risk. I’ve been looking for a new buy recommendation for months since the sale of the two 3-D printing companies. I think I’ve found it, in the form of a biotech company that went through about the worst times a biotech can have, survived to get its lead product to market, and is just hitting the ramp of rapid revenue growth. Yet it is still quite cheap, partly because there is a large short position based on a fundamental misunderstanding of its product’s position in the market.
Down’s Syndrome
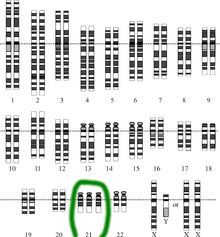
John Langdon Down first described Down’s Syndrome in 1866, to characterize what was a very common mental retardation. But it took almost 90 years before there was any molecular understanding of what causes Down’s Syndrome – an extra copy, or trisomy, of chromosome 21. Everyone has 23 pairs of chromosomes, with each pair made up of one chromosome from the father and one from the mother. But sometimes there is an extra copy – the trisomy – or a missing copy. The first publication about the link between Down’s Syndrome and trisomy was in the mid-1950s. Chromosome 21 is very small, with only 200 to 250 genes, which made it difficult to prove that this extra copy of chromosome 21 was the molecular underpinning of Down’s.
It took another 12 or 13 years before the first clinical amniocentesis was used to define chromosome 21, in 1968. So it’s only since the 1970s that we’ve an invasive procedure to fairly accurately find chromosome 21 trisomy. However, amniocentesis has a risk of miscarriage – 1 in 200 to 1 in 400, or 0.25% to 0.5%, depending on the institution.
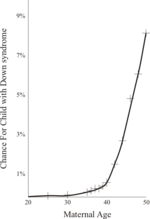
The risk of Down’s increases dramatically with the age of the mother:
At maternal age 20 to 24, the probability of a Down’s syndrome baby is one in 1,562. At age 35 to 39 the probability is one in 214, and above age 45 the probability is one in 19. Although the probability increases with maternal age, 80% of children with Down’s Syndrome are born to women under the age of 35, due to the overall fertility of that age group.
Recent data also suggest that paternal age, especially beyond 42, also increases the risk of Down’s Syndrome. My wife had our two daughters at age 40 and 45, when age 42 was just a fond memory to me. You can bet we were extremely interested in the outcome of her Down’s Syndrome screening, which we did with a blood test and ultrasound imaging of the baby’s neck.
Each year about 6,000 babies in the United States are born with this condition. Approximately 95% of these are trisomy 21. Down’s Syndrome occurs in all ethnic groups and among all economic classes.
Since the 1970s, several non-invasive serum tests have been developed, but they are not as accurate. As many as 275,000 women in the United States are falsely diagnosed as being at risk for carrying a child at risk for Down’s, and proceed to an amniocentesis. With a more accurate early test to rule out Down’s, many of these mothers could be spared further invasive tests. Clearly, what was needed was a better non-invasive test that is close in accuracy to the invasive procedure.
There are about 4.3 million births a year in the U.S., including 750,000 high-risk births – women over 35 (610,000), or with a family history of Down’s or another factor. About one in every 691 births is a Downie, according to the CDC. Although their IQs may be as low as 50, with love and attention many Downies have graduated from high school and held jobs – a very few even graduate from college. Their life expectancy is lower than normal and they tend to get serious diseases.
At around $1700 a test, this is currently a billion-dollar market in the U.S. alone. Many doctors think a non-invasive test should be extended to women over 25, which would roughly quadruple the available market.
Sequenom
Sequenom is a Torrey Pines, California company founded in 1994 to produce a gene sequencer, the MassARRAY DNA analysis system. They went public in March 2000 at $26, and the stock more than tripled the first day. It eventually hit $171.63. Although the MassARRAY system is still used in some research laboratories, Illumina and Affymetrix won that market. In 2005 the CEO resigned, they laid off 30% of the workforce, and turned to producing genetic tests available only to doctors, an important point discussed later, and focused on screening for Down’s Syndrome.
Some research at Oxford University and the Chinese Univeristy of Hong Kong showed that the DNA of a fetus circulates in the mother’s blood. Sequenom licensed the technology and set to work. By 2008, they showed very positive clinical trial results, and in January 2009 they released more positive results from a second clinical trial and said they would launch the test by July. The stock went over $20.
On April 24, 2009 the company cut 30 jobs due to falling sales of its MassARRAY system. Five days later, they postponed the launch of the Down’s Syndrome test because employees “mishandled” the study data that validated the product. In a few weeks, the stock fell to $3.53.
On Sept. 28, 2009 Sequenom’s CEO and the head of research were fired, and the CFO and another senior executive were forced to resign after an internal investigation. Three other lower-level people also walked the plank.
The Chairman of the Board, Harry Hixson Jr., took over as CEO. He was the former president and chief operating officer of Amgen. Before that, he was at Abbott Laboratories, first as Vice President, Research and Development, in the Diagnostics Division, and then as Vice President, Diagnostic Products Business Group. He brought a lot of credibility to a company that badly needed it. He was able to recruit senior-level people to the scientific advisory board.
In January 2010, Sequenom paid $14 million to settle a class-action shareholder lawsuit over the test data scandal. In June 2010 the former head of research, Elizabeth Dragon, pled guilty to conspiracy to commit securities fraud as part of an agreement with federal prosecutors. She cooperated with their investigation, and died eight months later, before she went to jail.
In February 2010, the prestigious American Journal of Obstetrics and Gynecology published clinical trial results showing that the Sequenom test detected all 39 blood samples from women who were pregnant with babies with Down’s Syndrome.
However, the new management decided to do a big trial to have enough impact to change the way doctors thought about screening for Down’s. They engaged the Women and Infants Hospital in Providence, R.I., to study the accuracy of the Down’s Syndrome test on more than 2,000 samples taken from pregnant women in the United States and nine other countries.
This was a clinical study that succeeded very well. The test showed a sensitivity of 98.6% in detecting Down’s Syndrome – it missed two of 212 cases. It had a specificity of 99.8%, which means it had one false positive. Both sensitivity and specificity had very tight confidence intervals. The results of this independent multi-center study demonstrating the performance of the test were published online on February 2, 2012 in the journal Genetics in Medicine.
In practice, doctors draw two vials of blood and send them to the Sequenom Center for Molecular Medicine, which is Sequenom’s lab. The fetal DNA is extracted and analyzed, and the company has found that having the second vial of blood lets them complete most tests without having to ask for more blood – a low “call rate” due to testing failure or low fetal DNA that would make that woman’s test indecisive and valueless.
Before Sequenom’s MaterniT21 test, even the best non-invasive screens had a detection rate of 90% to 95%, with a false positives rate of 2% to 5%. The MaterniT21 test detected Down’s Syndrome based on fetal DNA in a sample of the mother’s blood in 209 of 212 cases (98.6%). The International Society for Prenatal Diagnosis labeled MaterniT21 an advanced screening test in high-risk cases, based upon existing screening strategies. Of course, it has no risk of miscarriages. They pointed out that while it is very effective in diagnosing Down’s Syndrome, it currently cannot assess some other conditions which can be detected by amniocentises. MaterniT21 was rebranded as MaterniT21 PLUS in Februay of this year, and now detects trisomys at chromosome 13 (Patau Syndrome) and chromosome 18 (Edwards Syndrome). This quarter, it will begin determing fetal sex.
So this is not an investment in one test, it is an investment in a platform technology. Sequenom’s technology can be applied to a number of other diseases with a know genetic mutation cause. Tests for cystic fibrosis and age-related macular degeneration were introduced in 2011. The company also has researched tests to detect genetic material from malignant tumors circulating in the body, which might help oncologists diagnose cancer at an earlier stage, even before the tumor itself is detected.
The FDA
So far, the FDA has not required clinical trials for laboratory-developed genetic tests (LDTs) used by the same laboratory that developed it and offered only to doctors, not to patients. According to Sequenom’s 10-K:
“Historically, the FDA has exercised enforcement discretion and exempted from regulation LDTs created and used by the same laboratory. During a public meeting held in July 2010, the FDA explained that it was reconsidering its policy of enforcement discretion over LDTs. Citing a variety of safety concerns related to current LDTs, the FDA noted that the tests have become increasingly complex and utilized for significant medical decisions, sometimes in place of similar tests that have been reviewed and cleared or approved by the FDA. However, no formal guidance has yet been issued discussing the nature of the changes the FDA may make with respect to the regulation of LDTs, nor the scope of potential regulation. We continue to monitor potential changes as the FDA’s LDT policy evolves to ensure Sequenom CMM’s activities are consistent with the FDA’s most current policy.
“As part of the FDA’s evolving position on the regulation of LDTs, the FDA issued letters to a number of companies in mid-2010 that primarily related to direct-to-consumer genetic testing. In these letters, the FDA expressed concern about consumers making medical decisions in reliance upon genetic tests that have not undergone the FDA’s premarket review. Although Sequenom CMM does not sell its testing services directly to consumers, we also received a letter from the FDA in July 2010. We responded to the FDA by letter in August 2010 and met with the FDA in September 2010. We reiterated at that meeting that Sequenom CMM’s LDTs are physician-ordered and neither we nor Sequenom CMM are involved in direct-to-consumer commercialization. The FDA indicated at that time it had no further questions on the direct-to-consumer issue.”
Of course, the FDA only has jurisdiction in the United States, and there are other countries in the world that will purchase and use this test. Last week, LifeCodexx, Sequenom’s European licensee, got approval to offer testing services in Germany, Austria, Switzerland and Liechtenstein. Just this week, Sequenom announced international distribution agreements covering Japan, Hong Kong, the Czech Republic, Slovakia, the Netherlands, and Israel. These approvals and agreements increase the available market by another 500,000 women.
Also, Sequenom is going ahead with plans to file for FDA approval of its diagnostic tests in 2013, which will allow them to sell the tests as FDA cleared in vitro diagnostic (IVD) kits to other laboratories, and protect them if the FDA ever decides to regulate laboratory-developed tests (LDT) as in vitro diagnostics, even if they are sold only to doctors.
Marketing Results to Date
Sequenom introduced MaterniT21 in October 2011, becoming first-to-market with a genetic test for Down’s Syndrome. Their initial forecast was for 25,000 tests in 2012, and they raised that to 40,000 in April. On July’s earnings conference call, they said they have already done over 20,000 screenings, and raised their forecast to 50,000 tests by the end of the year – double the original estimate. They did 13,000 tests in the June quarter alone. In the next three years, I expect genetic screening to completely replace serum testing, ultrasound and amniocentesis as the easy first choice for risk-free, accurate Down’s Syndrome testing. Sequenom should do 100,000 tests in 2013, 200,000 in 2014 (their current capacity), and 400,000 in 2015.
The company has $98.6 million in cash compared to $20 million in debt. They reported a $29.6 million loss in the June quarter, 26 cents a share, on $18.3 million in sales. Revenues this year should hit $85 million, up 52% from 2011, followed by 88% growth to $160 million in 2013 regardless of what the economy does. The company should turn profitable in 2014 and grow profits rapidly thereafter.
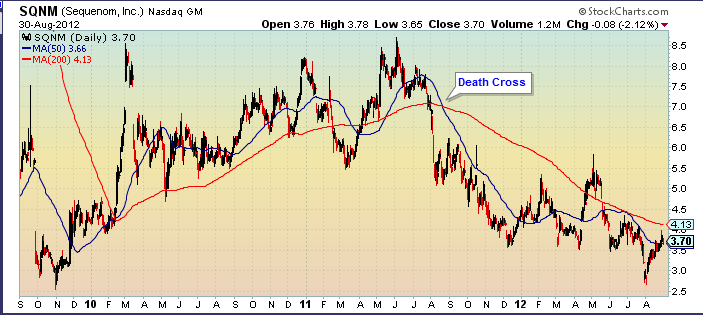
The stock closed today at $3.70, giving the company a $424 market capitalization. The 52-week range has been $6.22 to $2.65:
After setting a three-year low in early August, SQNM has regained the 50-day moving average and is headed for the 200-day moving average. Before the end of the year I expect the 50-day to break through the 200-day – the “Golden Cross” that will terminate the “Death Cross” from August 2011.
SQNM was cut in half from May to early August by two small setbacks, and worries about competition. First, they missed the June quarter consensus estimates for $19.1 million and 21 cents a share. Right now, Sequenom is accounting on a cash basis until they get some experience with the percentage of reimbursement they can expect when they do a test. That means the expenses hit right away, but the revenues trickle in over the next six months as they are reimbursed. A small miss is meaningless, because the consensus is a WAG.
Second, Coventry Health had agreed to pay for MaterniT21 tests beginning July 1, and then changed their mind and said they will terminate coverage on August 31. MultiPlan, a huge payer with 55 million members and 900,000 providers, still covers the test. Medicare does not, of course. I expect most European health plans to cover the test wherever it is approved. In the U.S., the company will have to get commitments to cover from one insurer at a time.
Third, there are two private competitors, Ariosa Diagnostics (formerly Aria Diagnostics) and Verinata Health. Each plans to launch a genetic test for Down’s Syndrome within a year. (Sequenom is suing Ariosa for patent violations, and Verinata is suing Sequenom.) Although Wall Street is worried about the potential competition, recent data from each of these companies shows a call rate of 4.6% for Ariosa and 6.9% for Verinata. As I mentioned above, the “call rate” is due to testing failure or low fetal DNA that makes the test indecisive and valueless. Sequenom has a call rate of 0.8%, and is already in the market with a 75-person sales force and a marketing program.
Ariosa and Verinata probably will offer their lower-quality tests at a lower price. But Sequenom has reduced the cost of doing the test to around $500, and will be under $300 in a year. They are in a good position to lower the price to doctors enough to make Ariosa and Verinata also-rans. This also ties into the question of insurance reimbursement, as Sequenom has a lot of latitude with private insurers to trade an agreement to cover for a discount on the cost of the test.
Selling for only 2.7x next year’s revenues, which should grow 60% to 80% a year for the next few years, with 23% of its market capitalization in cash, SQNM is cheap. Management bought 170,000 shares for themselves in July – these were not options grants. The CEO bought 50,000, the CFO bought 10,000, the Chief Accounting Officer bought 10,000 and the Director of R&D bought 100,000. Short interest of 31.7 million shares is 28% of the outstanding shares. At recent trading volumes, it would take the shorts 10.4 days to cover, buying every share.
The Bottom Line: SQNM has long-term options, but at these low levels I think it is smarter to buy the stock. Fed Chairman Bernanke is speaking tomorrow at 10:00am ET. I would not buy SQNM until he is done. If he disappoints the market, I think you will be able to get SQNM under $3.50 a share. If he announces QE to infinity, the market will rally sharply, but it will be industrial and commodity stocks that lead it up. You still should be able to get SQNM under $4 easily. I want you to wait for Bernanke, and then about 15 minutes after his remarks become public (they may be released early) you should buy SQNM anywhere under $4 for an $8 target in 2013 and $12 in 2014. I am making the stock a Trading Buy for continued good news on the MaterniT21 PLUS rollout. I expect to make it a core holding sometime next year, after I can assess the competitive situation.
Market Outlook
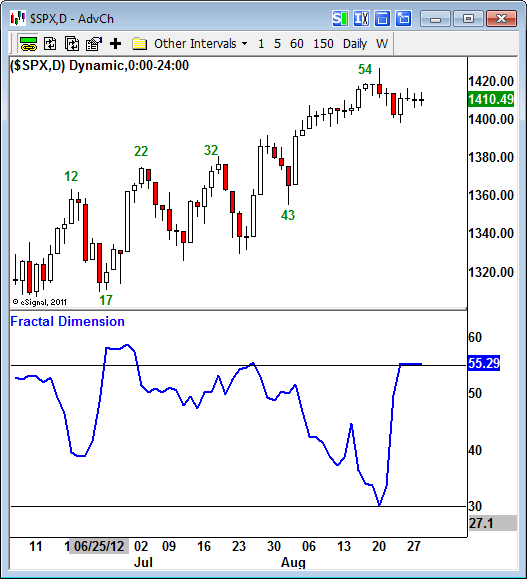
The SP 500 has been in a very tight holding pattern waiting for Bernanke’s speech – a “coil.”
Coils often break in a false move up or down, and then quickly reverse to reveal the true direction of the next move. This is something to watch carefully tomorrow.
Coming Events
All times below are ET, and most of the presentations and slides are archived on the companies’ websites so you can listen to them. Seems to be ARNA and DNDN week,
Wednesday, September 5
ARNA – Arena Pharmaceuticals – Unspec. panel presentation – CitiCorp 7th Annual Biotech Day
INFN – Infinera – 2:15pm – Citi 2012 Technology Conference
DNDN – Dendreon – 3:50pm – Stifel Nicolaus Global Healthcare Conference
Thursday, September 6
ATRS – Antares Pharma – 8:35am – Stifel Nicolaus Global Healthcare Conference
ARNA – Arena Pharmaceuticals – 9:10am – Stifel Nicolaus Global Healthcare Conference
DNDN – Dendreon – 12:50pm – Baird Health Care Conference
Friday, September 7
ARNA – Arena Pharmaceuticals – 10:30pm – NewsMakers in the Biotech Industry
The Recovery Trade Sequence: Tech, Biotech & Energy
Arena Pharmaceuticals (ARNA) will be at three brokerage firm conferences next week, which should support the stock. ARNA is a Core Holding and a Trading Buy under $10 for the European/ROW partnership announcement and Belviq rollout, with a $22+ target in 2012.
The Recovery Trade Sequence: Gold & Silver
Paramount Gold & Silver (PZG) will move with the precious metals (see below), where a major Intermediate Cycle buying opportunity is at hand. This probably will be the biggest cycle of the new C-wave as it plays out into 2014. PZG is a Core Holding and a Trading Buy under $4 for my $15 target when the company is acquired, possibly in 2012. There is a lot of good news coming.
Major News Coming to Drive Our Stocks
This list has its own page. I’ll comment on any changes here, and you can always click the link under Member Resources. There are no changes this week.
Biotech MegaShift
Dendreon (DNDN) is determined to convince urologists and oncologists to sequence Provenge treatment first, followed by Johnson & Johnson’s Zytiga, as “best practice” for treating hormone-refractory prostate cancer patients. The FDA just granted priority review of Zytiga in pre-chemotherapy patients, the population Provenge is approved for. I’m sure the usual shorts will be out with their FUD that this will kill DNDN, ignoring the powerful benefits of sequencing.
In the field, Zytiga’s momentum is stalling. Currently, about 20% of doctors say they use Zytiga instead of Provenge but that’s down from earlier surveys showing 31%. I have not seen a single survey of how many doctors are sequencing the drug, even though that is the current recommendation of the National Comprehensive Cancer Network and was seconded by The Journal of Clinical Oncology.
The reasons are very simple. Patients have to be steroid-free to take Provenge over a period of six weeks. Zytiga has to be given with steroids for 13 months. Provenge shows a survival advantage at six months. Zytiga does not show a survival advantage until 18 months, several months after the therapy ends. Who would not use Provenge first?
DNDN is a Core Holding and a Trading Buy that I would buy up to $7 short-term, with a $12 target after a European partner is announced. Longer term, I would buy all the way up to $28, with a $50 target as Provenge revenues accelerate and they file for approval in Europe.
Zalicus (ZLCS) should see increased Exalgo royalties from the FDA approval of a 32-milligram dose. Covidien said it will launch the new version of Exalgo in the next few weeks. Since Exalgo is the lead drug for the pharmaceutical company they plan to spin-out, I expect them to make a big push on this stronger formulation.
Zalicus filed a $75 million shelf registration – this is not a current offering of stock or bonds. With the Synavive results due soon, I am moving ZLCS to a Trading Buy, still under $2 for my $7 target in mid-2013.
Content on Demand MegaShift
DragonWave (DRWI) is benefitting from the now-rapid deployment of Sprint’s Network Vision 3G/4G network. According to a CTIA survey of wireless customers, wireless data traffic increased 123% from 2010 to 2011. There is just no way the carriers can avoid deploying more and faster spectrum, and moving on to Internet Protocol microwave backhaul for many of those towers. DRWI is a Core Holding and Trading Buy under $6 for a $20 target in 2013.
QuickLogic (QUIK) should benefit from the introduction of General Imaging’s ipico picoprojector for the holiday season. The ipico is based on Micron’s PopVideo product, which basically is a reference design for selling Micron displays. Micron recently sold the PopVideo product line to the Japanese company, Citizen. I don’t know what Citizen will do with it, but Micron is not a consumer products marketing company, while General Imaging is superb in that area.
As you can see from their website, General Imaging supplies the GE digital cameras to retailers, and is making a big splash with the ipico that includes QUIK’s VEE and DPO chips. Their picoprojector has triple the light output of the PopVideo. You can read about it at BeABigShowoff.com.
The net is that I expect a much stronger holiday season than I had been thinking, and a faster ramp of sales to a much higher level than we would have seen with only Micron marketing a picoprojector. This will be a major driver of QUIK’s fourth quarter results, as well as in 2013. QUIK is a Core Holding and a Trading Buy up to $5 buy limit for my $12 target in 2012.
Sprint (S) added four more cities to its 4G network, as they race to be ready for Apple’s iPhone 5 introduction on September 12. S is a Value Buy under $4 for my $8 target in 2012 – which I expect to raise.
Towerstream (TWER) said in its last conference call that it was starting a WiFi trial with a national news media company. Now we know it is the Wall Street Journal, which is offering free WiFi to its subscribers in New York City and parts of San Francisco. An article in The Verge broke the story. TWER is a Core Holding and a Trading Buy up to $5 for a $9 target in 2012. It will take to 2013 to get their top 20 markets all open and profitable, and then I expect it to hit my $16 target
Hyperinflation MegaShift
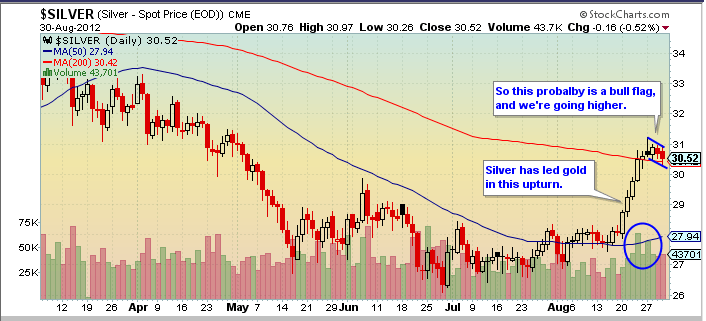
Silver is leading gold up in this very strong daily cycle. The next dip to a daily cycle low in October or so should also mark an intermediate cycle low, to be followed by a very big rally in this new C-wave. C-waves always chop around for a while, building a base, as we have seen so far in 2012. Then, when they’ve thrown off all the bulls and trapped as many bears as possible, they head for the moon – giving those on the sidelines no easy opportunity to get in.
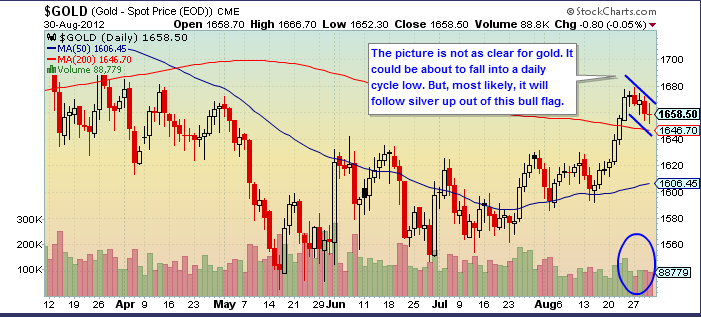
But in the short-term, sentiment has gotten awfully bullish. I suspect the current bull flags will break to the upside, power gold to $1700, and then trap all the new bulls.
Silver:
But silver sentiment:
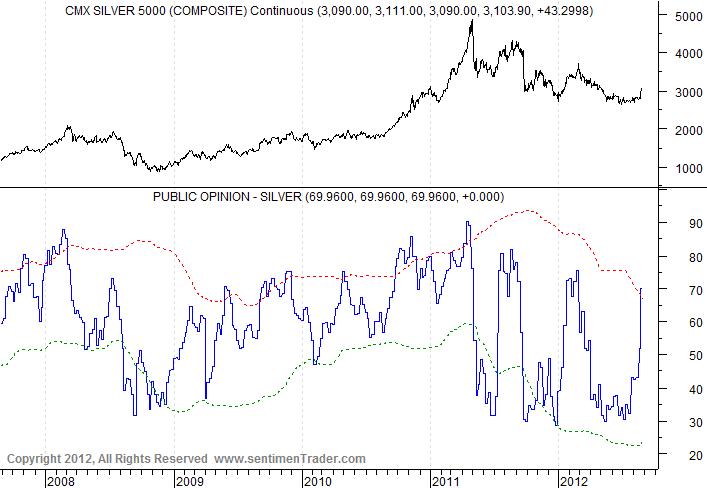
Gold is doing well, although not as well as silver:
But gold sentiment also is too high:
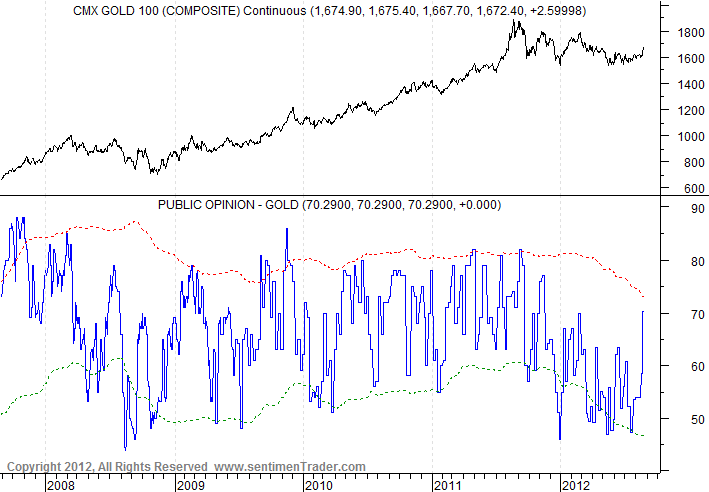
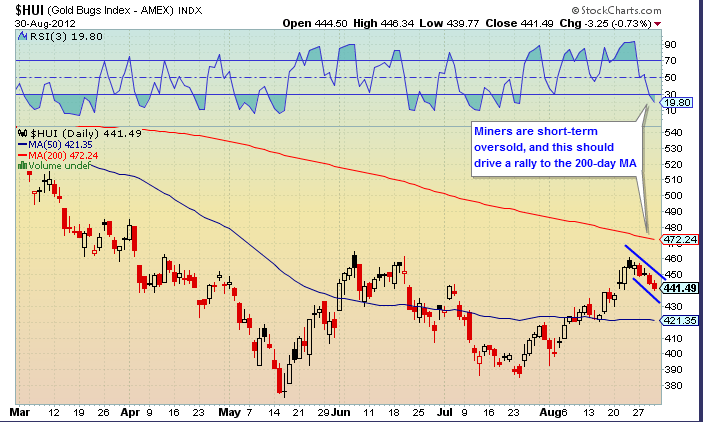
And the miners still have some room to run, before a takedown into the Intermediate Cycle low:
Energold (EGD.V; EGDFF.PK on the pink sheets) missed the consensus earnings expectation for the June quarter due to mineral drilling activity decreasing 26% as a result of a cautious drilling environment among junior miners. As I expected, the juniors have “disappeared from the marketplace” due to the weak capital market conditions, affecting their ability to raise capital to fund exploration programs. The company said they also had politically-based slowdowns in two South American countries, including Peru. Although the indication is that those slowdowns are over, the company has redeployed rigs to other markets that will begin generating revenue again over the next two quarters.
They did $32.7 million in revenues and reported a four cents per share loss, versus a consensus for $32.7 million and a nine cent profit. The stock dropped 10% after they reported yesterday to $3.18, but snapped back 22 cents today to close at $3.40. EGD.V is a Long-Term Buy under $4.50 for a $10 target price in 2012.
New Energy Technology MegaShift
Axion Power (AXPW) will be a participant at the 13th European Lead Battery Conference in Paris in September. The focus will be microhybrids vehicles with stop-start idle elimination, a technology that will be used in 35 million cars a year by 2015. All the major car companies and battery manufacturers will be there, and be exposed to Axion’s lead-carbon solution to building a battery that can stand up to start-stop requirements. AXPW is a Core Holding to buy under $1.00. My 2012 target is $1.50, and I think this will be a 10-bagger over the next few years.
The Geothermal Basket – Top Buy Long Term
I recommend putting 50% of your basket into ORA, with 25% each in the two smaller companies: AXY.TO and HTM.
Ormat Technologies (ORA) got $310 million in limited-recourse project financing from the Overseas Private Investment Corporation (OPIC), an agency of the U.S. Government. They’ll use the money to build the Olkaria III geothermal power complex located in Naivasha, Kenya.
Hey, wait a minute – OPIC reports to President Obama – a Kenya project – surely somebody can make something out of this in an election year! (Kidding, kidding.) ORA is a Long-Term Buy up to $25 for my $40 target in 2012.
* * * * *
Action Items This Week
* * Buy SQNM after Bernanke speaks tomorrow
* * Buy ZLCS for the Synavive trial results coming soon
* * Buy QUIK for a much-improved outlook for picoprojector sales this year
* * * * *
The U. S. hit $16 trillion in debt this week, continuing its record run as the greatest debtor in the history of the world. It took over 200 years to accumulate the first trillion dollars of debt. It took only 286 days to accumulate the most recent trillion dollars of debt. At an average interest rate of 2.13%, the budget is $340 billion in interest payments this year. China owns so much U.S. debt that their interest income from the Treasury Department almost covers their entire military budget.
The next trillion will be here before you know it. The trend is unsustainable, but Obama and Romney are arguing about very different visions of how to borrow money, shift it around, and not pay it back. [Credit:Bloomberg Business News] I hope Gary Johnson picks Ron Paul as his Veep, if only to shake up the game.
* * * * *
Your trying to smile during a Presidential election Editor,
Michael Murphy, CFA
Founding Editor, New World Investor
The Core Holdings
Arena Pharmaceuticals (ARNA)
Antares Pharma (ATRS)
BioCryst Pharmaceuticals (BCRX)
Dendreon (DNDN)
Rochester Medical (ROCM)
DragonWave (DRWI)
QuickLogic (QUIK)
Towerstream (TWER)
A Short-Sale or REO House
A Bag of Junk Silver
Market Vectors Gold Miners Exchange-Traded Fund (GDX)
Market Vectors Junior Gold Miners Exchange-Traded Fund (GDXJ)
Paramount Gold & Silver (PZG)
Sprott Resource Lending (SILU)
Axion Power (AXPW.OB)
The Long-Term Buys
Aastrom Biosciences (ASTM)
Energold (EGD.V; EGDFF.PK on the pink sheets)
Golden Predator (GPD.TO; GPRXF.PK on the pink sheets)
Otis Gold (OOO.V; OGLDF.PK on the pink sheets)
Infinity Energy Resources (IFNY.PK)
Alterra Power (AXY.TO; MGMXF.PK on the pink sheets)
Ormat Technologies (ORA)
US Geothermal (HTM)
The Value Buys
Sprint (S)
Primero Mining (PPP; P.TO in Canada)
Exide (XIDE)
The Trading Buys
Arena Pharmaceuticals (ARNA) – for the European and ROW partnerships, and launch of Belviq
BioCryst Pharmaceuticals (BCRX) – for the BCX4208 gout drug parthership announcement
Dendreon (DNDN) – Continued roll-out of Provenge; possible takeover bid from Roche
NEW!!! Sequenom (SQNMN) – Continued roll-out of MaterniT21 PLUS Down’s Syndrome test
MOVED!!! Zalicus (ZLCS) – Synavive trial results in September or October
DragonWave (DRWI) – IP microwave backhaul taking off; big contract wins coming; Nokia Siemens microwave business acquisition
QuickLogic – Consumer demand for tablets and smartphones with VEE & DPO chips
Towerstream (TWER) – Leading supplier of wireless business telecom services hits cash flow breakeven next; more WiFi offload announcements coming shortly
Market Vectors Gold Miners Exchange-Traded Fund (GDX) – the big miners
Market Vectors Junior Gold Miners Exchange-Traded Fund (GDXJ) – the smaller miners
Paramount Gold & Silver (PZG) – Sleeper preliminary economic report coming, and a 43-101 on San Miguel
Infinity Energy Resources (IFNY) – partner news and 3D seismic program
Rentech (RTK) – high corn prices and low natural gas prices for another year
Holds
Connacher Oil & Gas (CLL.TO; CLLZF.PK on the pink sheets)
Harmonic (HLIT)
Harris & Harris (TINY)
Infinera (INFN)
Ocean Power Technologies (OPTT)
© Copyright – Next Paradigm Press – 2012. New World Investor does not act as a personal investment adviser or advocate the purchase or sale of any security or investment for any specific individual. The recommendations and analysis presented to members is for the exclusive use of members. Members should be aware that investment markets have inherent risks and there can be no guarantee of future profits. Likewise, past performance does not assure future results. Recommendations are subject to change at any time. Nothing in this presentation should be considered personalized investment advice. No communication to you by Michael Murphy or any of our employees or contractors should be deemed as personalized investment advice.New Post for Co
 Print This Post
Print This Post